Benchmark Dose Modelling in Chemical Risk assessment
In this guide
In this guideIdentifying a Reference Point from Toxicity studies
10. Hazard characterisation is a key step in the risk assessment pathway. It attempts to establish the nature and severity of (an) adverse effect(s) associated with exposure to a chemical, with particular attention paid to the relationship between the dose and effect (COT, 2020). Toxicity studies, carried out to characterise these adverse effects, are typically designed to identify a dose that can be used as a starting point for human health risk assessment. This dose is often referred to as the RP or the Point of Departure (PoD) (COT, 2020; EFSA, 2009).
11. Traditionally, RPs have been determined using the (NOAEL) or (LOAEL). The NOAEL (historically also sometimes referred to as a No Observed Effect level, NOEL) is a means of establishing a RP by determining the highest dose of a substance at which no (statistically) significant adverse effects are observed (FAO/WHO, 1990). While some variation exists in the statistical approaches, determination of the NOAEL typically involves multiple pairwise comparisons of the data at different doses, to an appropriate control data set. This approach can be used for data types including continuous data (i.e. data measured on a continuum, e.g., organ weight or blood biomarker concentration) or Dichotomous Data, also known as Quantal Data (i.e. Data where an effect may be classified into one of two possible outcomes, e.g., dead or alive, with or without incidence of a specific symptom such as tumours). Where statistically significant effects are detected at all dose levels tested, the lowest dose used in the study (i.e. the LOAEL) may be selected as the RP. In this case additional uncertainty factors are often recommended if the RP is used to produce a corresponding HBGV, in recognition of the fact that a lower dose may still cause an adverse effect. Conversely, if no statistically significant effect is observed at any of the dose levels, the highest dose is typically selected as the NOAEL (EFSA, 2022).
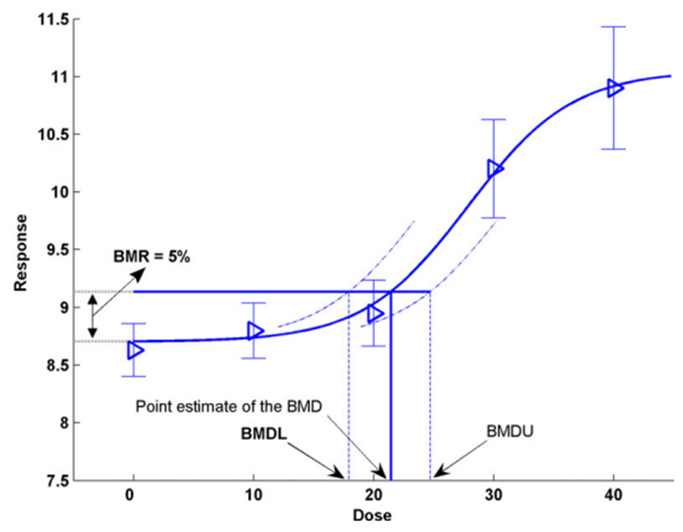
12. An alternate to the NOAEL/LOAEL is the BMD approach (Crump, 1984). The BMD is a dose level, estimated from a fitted dose-response curve or curves, associated with a pre-specified change in response (the benchmark response, BMR) relative to the control group (background response). Instead of comparing individual groups (doses), the BMD approach considers all the available dose-response data to estimate the shape of the overall dose-response relationship for a particular endpoint (Figure 1). It is possible to derive confidence levels of the BMD response from the dose-response modelling with the BMD lower confidence level (BMDL) typically taken as the RP for establishing HBGVs (EFSA, 2022).
Figure 1. Illustration of the BMD approach using hypothetical continuous data (Figure taken from EFSA, 2017). Hypothetical experimental mean response data (triangles) are plotted along with their confidence intervals. The solid curve represents the fitted dose-response model. The curve determines the point estimate of the BMD, generally defined as a dose that corresponds to a low but measurable change in response, and here representing a benchmark response (BMR) of 5%. The dashed curves represent, respectively, the upper and lower 95% one-sided confidence bounds for the effect size as a function of dose. Their intersections with the horizontal line are at the lower and upper bounds of the BMD, denoted BMDL and BMDU, respectively.
13. Both the U.S. EPA and EFSA now recommend using the BMD approach, where appropriate, as the preferred means to identify a RP for deriving a HBGV. This is also the stated preference of the JECFA and JMPR (Joint FAO/WHO Meeting on Pesticide Residues).