Scope of the Nutrition and maternal health project
On this page
Skip the menu of subheadings on this page.This is a paper for discussion. This does not represent the views of the Committee and should not be cited.
Introduction
1. The Scientific Advisory Committee on Nutrition (SACN) last considered the maternal diet and nutrition in relation to offspring health in its reports on ‘The influence of maternal, foetal and child nutrition on the development of chronic disease in later life’ (SACN, 2011) and on ‘Feeding in the first year of life’ (SACN, 2018). In the latter report, the impact of breastfeeding on maternal health was also considered. In 2019, SACN agreed to conduct a risk assessment on nutrition and maternal health, focusing on maternal outcomes during pregnancy, childbirth and up to 24 months after delivery.
2. SACN agreed that, where appropriate, other expert committees would be consulted and asked to complete relevant risk assessments. In 2020, a scoping paper was presented to the COT (TOX/2020/45) to define the scope of the work from a toxicological safety perspective and request their input on the selection of candidate chemicals or chemical classes that could be added or removed.
3. In 2024, the COT recommended drafting an overarching Annex for the maternal diet and nutrition project to define the reproductive and developmental life stages under consideration, the endpoints for assessment, and the list of chemicals of interest. This Annex (COT/2025/44), incorporating initial discussions, was presented to the COT in December 2025.
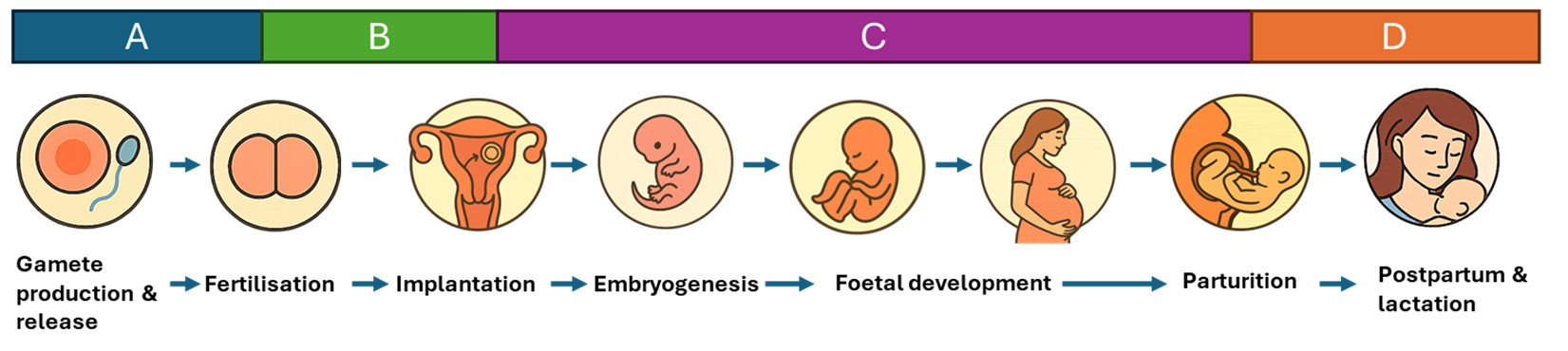
4. The COT requested that the schematic outlining the reproductive and developmental life stages in COT/2025/44 would be more appropriately represented as a linear rather than circular diagram to better reflect the scope of the SACN project. The schematic has therefore been revised to a linear format.
5. The FSA Secretariat, in consultation with the SACN Secretariat, revisited the scope of the SACN maternal diet and nutrition work to confirm the relevant endpoints for this project. The SACN Secretariat advised that the COT assessments should focus on maternal outcomes during pregnancy, childbirth, and up to 24 months postpartum; effects on the embryo and foetus are also in scope. Whilst the 24‑month post-partum period applies specifically to maternal outcomes, there may be instances where neonatal effects may also be considered relevant. Paragraph 3 of Annex A sets out the endpoints proposed in the SACN scope
Questions for the Committee
6. The Committee are asked to consider the following questions:
a) Does the Committee have any comments on the schematic presented in Figure 1?
b) Does the Committee have any further comments on the proposed endpoints and list of chemicals for consideration?
c) Does the Committee have any other comments on the Annex?
Annex A to TOX/2026/10
1. In 2019, Scientific Advisory Committee on Nutrition (SACN) agreed to conduct a risk assessment on nutrition and maternal health focusing on maternal outcomes during pregnancy, childbirth and up to 24 months after delivery. As part of this project, SACN sought the advice of the COT on whether excess exposure to a variety of chemicals could pose a risk to maternal health. Chemicals are only to be covered if they are related to a toxicological mechanism rather than inadequate nutrition. This Annex contains information on the scope of SACN maternal health and nutrition project, the proposed endpoints and the current list of chemical contaminants and nutrients to be assessed by the COT.
2. As illustrated in Figure 1 below, the following life history stages in the mother will be assessed: preconception (stage A), pre-implantation (stage B), pregnancy (stage (C) and post-partum, including lactation (stage D). SACN has considered preconception (stage A) from a biological perspective, defining it as the critical period spanning the days to weeks before embryo development. However, the COT may wish to define this period more precisely for the purposes of this project. The pre-implantation (stage B) is defined as the time from conception to implantation, while pregnancy (stage C) covers the period from implantation to parturition. The post‑partum period (stage D) includes any maternal outcomes occurring up to 24 months after delivery, including adverse effects on lactation. However, it does not include effects on the offspring that are mediated through lactation. More broadly, this stage excludes the growth, development and sexual maturation of the offspring, as these were considered outside the scope of the project. Infant outcomes may be considered in specific instances, but only where they relate to the neonatal period (i.e. the first few days after birth).
Figure 1: The maternal life history stages, including stage A (pre-mating to conception), B (conception to implantation), C (implantation to parturition), D (post-partum including lactation).
Proposed endpoints
3. A range of maternal outcomes during pregnancy, childbirth and up to 24 months after delivery will be considered, aligning with the range of relevant endpoints identified by SACN to be examined from a toxicological perspective, with further discussion needed on any additional outcomes the COT may regard as relevant.
Pregnancy related outcomes, including:
- Time to conceive,
- Congenital anomalies (COT expanded to this to include other adverse effects on embryonic and foetal development),
- Miscarriage,
- Stillbirth,
- Preterm delivery,
- Gestational age at delivery,
- Birthweight and size for gestational age,
- Perinatal and neonatal morbidity and mortality,
- Placental health.
Maternal health outcomes, including:
- Blood pressure and hypertensive disease in pregnancy, including the risk of gestational hypertension and pre-eclampsia.
- Mental health (when clearly linked to chemical exposure),
- Glycaemic control, including risk of gestational diabetes,
- Anaemia,
- Bone health, including maternal bone mineral density and bone mass,
- Effects on lactation but not mediated by lactation.
The following endpoints were considered to be outside the scope of the COT
- Mode of delivery,
- Oral health,
- Weight loss, weight gain and weight retention during pregnancy (gestational weight) and up to 24 months after delivery.
Proposed list of chemicals for consideration
4. The proposed chemicals to be considered by the COT are discussed below. Chemicals will only be included where their effects are associated with a toxicological mechanism rather than inadequate nutrition. It should be noted that the list of chemicals may be subject to change, with new additions made where SACN and the COT consider this appropriate.
Vitamins and minerals
- Vitamin A, C, D (including calcidiol), E,
- Selenium, iodine.
Heavy metals
- Lead, arsenic, mercury, cadmium. This was subsequently expanded in include a more general consideration of pica behaviour.
Other food components/herbal supplements
- Tea (Green and black), caffeine, ergot alkaloids, raspberry leaf, echinacea, camomile, peppermint, evening primrose oil, dandelion, resveratrol, phytoestrogens.
Dietary contaminants
- Mycotoxins: citrinin, aflatoxins, fumonisins, ochratoxin A, Zearalenone, deoxynivalenol, nivalenol, patulin, T2 and HT2,
- Oily fish.
Process contaminants
- Acrylamide, heterocyclic amines.
Organic contaminants
- Dioxins and dioxin-like polychlorinated biphenyls PCBs,
- Non-dioxin-like (NDL) PCBs,
- Bisphenol A (BPA),
- Pesticides/legacy pesticides.
Completed papers
5. The below table list the chemicals that have been reviewed by the COT and have published final statements.
Table 1. List of chemicals with completed discussion papers and statements published by the COT before the publication of this Annex A (Scope of the Nutrition and maternal health project) *
|
Chemical |
Statement |
Link |
|
Vitamin D |
Statement on the potential effects of excess vitamin D intake during preconception, pregnancy and lactation |
|
|
Iodine |
Statement on the potential effects that excess iodine intake may have during preconception, pregnancy and lactation |
|
|
Vitamin A |
Statement on the effects of excess Vitamin A on maternal health |
|
|
Cadmium |
Statement on the potential risks from cadmium in the maternal diet |
|
|
Effects on Pica during pregnancy |
Discussion paper on the effects of pica during pregnancy |
TOX-2023-06 Discussion on the effects of pica during pregnancy .pdf |
|
Lead |
Statement on the potential risks from lead in the maternal diet |
Statement on the potential risks from lead in the maternal diet |
|
Raspberry leaf |
Statement on the potential health effects of raspberry leaf tea in the maternal diet |
Statement on the potential health effects of raspberry leaf tea in the maternal diet |
|
Ginger |
Safety of Ginger Supplement Use in Pregnancy |
|
|
Ergot alkaloids |
Statement on the potential risks from ergot alkaloids in the maternal diet |
Statement on the potential risks from ergot alkaloids in the maternal diet |
|
Mercury |
Statement on the effects of mercury on the maternal health |
|
|
Citrinin |
Statement on the potential risks from citrinin in the maternal diet |
Statement on the potential risks from citrinin in the maternal diet |
*papers published before publication of Annex A may have slight deviations in the scope of endpoints assessed.
Table 2. List of chemicals with completed discussion papers and statements published with Annex A (Scope of the Nutrition and maternal health project)*
|
Chemical |
Statement |
Link |
|
Echinacea |
Statement on the potential health effects of Echinacea in the maternal diet |
To be published shortly. |
Secretariat
March 2026
References:
FDA (1966). Guidelines for reproduction studies for safety evaluation of drugs for human use. Washington, Department of Health, Education and Welfare. Referenced in: Raheja KL, Jordan A, Fourcroy JL (1988). Food and drug administration guidelines for reproductive toxicity testing. Reproductive Toxicology, 2(3-4), 291-293. https://doi.org/10.1016/0890-6238(88)90034-2
FDA (2021). S5(R3) Detection of reproductive and developmental toxicity for human pharmaceuticals. Guidance for industry. The International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH). Guidance for Industry
IPCS (2001). Principles for evaluating health risks to reproduction associated with exposure to chemicals. Environmental Health Criteria, 225. Principles For Evaluating Health Risks To Reproduction Associated With Exposure To Chemicals (EHC 225, 2001)
Spielmann H (2009). The way forward in reproductive/developmental toxicity testing. Alternatives to Laboratory Animals, 37(6), 641-656. https://doi.org/10.1177/026119290903700609
Spielmann H. The Way Forward in Reproductive/Developmental Toxicity Testing. Alternatives to Laboratory Animals. 2009;37(6):641-656. doi:10.1177/026119290903700609