Opportunities and outlook for UK Food and Chemicals regulation post EU Exit Workshop Report 2022
On this page
Skip the menu of subheadings on this page.https://doi.org/10.46756/sci.fsa.ebr546
Opportunities and outlook for United Kingdom Food and Chemicals regulation post European Union Exit- Workshop Report (2022)
Background and Objectives
1. In 2019, the Royal Society of Chemistry held a workshop: Royal Society of Chemistry (RSC) Workshop of 2019 : Drivers and scope for a UK chemicals framework. Presentations and discussions at this event examined chemical regulation in the United Kingdom (UK) post European Union (EU) exit and the opportunities that might be realised.
2. Since then, several global events have impacted the economy and regulation in the UK, including EU exit. Following these events, the Committee on the Toxicity of Chemicals in Food, Consumer Products and the Environment (COT) decided it would be timely to have a second workshop to build on the successful 2019 workshop by reviewing what has been achieved and what still needs to be done to realise the full potential of EU exit in the area of food and chemical regulation.
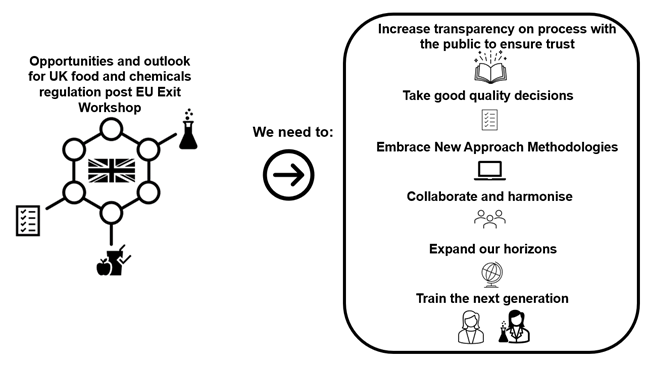
The purpose of the workshop was to review the food and chemical regulatory landscape; its transfer to the UK; future UK development of Registration, Evaluation, Authorisation and Restriction of Chemicals (REACH) and divergence (drivers and supporting science); identify the challenges and opportunities to consider where new structures as well as investment are required to realise and address these.
Workshop Overview
3. The workshop took place on the 13th of July 2022 in Liverpool, UK. Participants were from industry, academia and regulatory agencies. The day was divided into three sessions:
- The landscape of regulation post EU exit: UK stakeholder perspectives, International perspectives, opportunities and challenges for UK divergence;
- Major drivers for change and potential impact on chemical regulation; and
- Indirect Effects: food prices, food security, supply chain, fraud (Food regulation/human health).
4. Each of the sessions consisted of presentations followed by a roundtable discussion and included interactive sessions.
Introductions and aims of the day
Dr Phil Botham introduced the workshop and explored the theme of a time of transition.
5. There is currently the opportunity to think about what the new legislative framework could look like in the UK for chemicals and food safety, subsequent to EU regulation. Opportunities for the UK include for example, the earlier application of scientific developments, including New Approach Methodologies (NAMs). However, for these to be used and integrated there will need to be acceptance of their applicability from regulators, as well as public acceptance that they will continue to ensure the safety of food and chemicals. There is currently a relatively slow pace of transitioning to the use of such methods under EU legislation, such as EU REACH where it is unlikely that a significant transition will occur as part of the ongoing review of the data requirements. However, the European Food Safety Authority (EFSA) has indicated that it has the ambition to fully implement the use of validated NAMs in the safety assessment of food by 2027.
6. It was highlighted that the COT supports requests from the Food Standards Agency (FSA), the UK Health Security Agency (UK HSA) and other Government Departments across a wide range of chemicals, but its remit also includes considering the broader aspects of scientific developments. One possible outcome is to be more proactive in developing frameworks for the assessment of chemicals and contaminants in the UK.
7. Attendees were invited to use the workshop to explore the opportunities to further develop an advanced risk framework for food and chemicals regulation.
UK chemicals and food regulation: Where are we at post EU exit and what needs to be done?
Professor Michael Walker introduced the session with a review of the current process for regulated product authorisation within the UK. The UK currently has a unique pipeline for regulated products compared to when the UK was part of the EU process.
8. Legislation.gov.uk displays the current UK legislation. The site features ex-EU law incorporated into UK statutory law.
9. The potential for advantageous divergence of legislation between the EU and the UK was then introduced.
10. A key reason for divergence could be scientific opinion. The conclusions on scientific evidence reached by expert groups in the UK may be different to the opinions from within the EU. This might be because: expert groups are requested to answer a different question on a key topic, or they might be asked in a different way, or differences in membership of expert groups may be a factor.
11. Legislative divergence is to be expected; either in part or entirely. This can be driven by public opinion, the political climate and/or differences in legislative programmes. Applications for changes to regulated products that may vary in their content and timings between the UK and EU is another potential point of divergence. Local authority input would also potentially influence divergence. However, it was unlikely that local authorities would have the resources to achieve this.
12. In the context of the work of the Joint Expert Groups (JEGs), there have been occasions when it would have been beneficial for local authorities to inspect the analytical methods and processes undertaken by companies. To support the FSA’s Scientific Advisory Committees (SACs) as they provide independent scientific review of regulated products, the FSA had created three new JEGs for regulated products. These work to the same principles as the SACs. The COT, the Committee on Carcinogenicity of Chemicals in Food, Consumer Products and the Environment (COC) and Committee on Mutagenicity of Chemicals in Food, Consumer Products and the Environment (COM) will continue to provide risk assessment advice for risk-based standards and controls, while the JEGs will undertake most of the work required for regulated products. The three JEGs focus on: Additives, Enzymes, and other regulated products (AERJEG) Animal Food and Feed Additives (AFFAJEG) (now superseded by the Advisory Committee on Animal Feeding stuffs (ACAF), and Food Contact Materials (FCMJEG).
13. Non-targeted analysis was then discussed. This relates to substances that may be within a product that neither the manufacturer nor regulator know are going to be present. A large amount of the non-targeted analysis work is focused on food authenticity. Traditionally, analytical chemists have determined preselected chemical target species, using information collected from techniques that are hopefully robust and selective. It was stated, that in previous JEG work performance characteristics had been considered for a range of traditional chemical and bioanalytical techniques, to establish a view on what was fit for purpose. These performance characteristics are well known, and include: limits of detection and quantification, bias, recovery, and measurement of uncertainty. These exist within an internationally agreed framework of method validation. This is not the case for non-targeted analysis. Dr Michael Walker, along with FSA employees, JEG Members and an academic from Queens University, Belfast have been working on the issue of non-targeted analysis. This included creating the definitions presented below, to aid the construction of a common language surrounding non targeted analysis.
Targeted method: A targeted method produces information on the concentration of a predetermined, selected, component from one of its characteristic signals.
Non-targeted method: A non-targeted method produces information on a component or components, not defined a priori, using a chemometric examination of multiple parameters, using a large database of parameters from all potential components for identification.
A partially non-targeted method: A partially non-targeted method produces information on components in a sample after chemometric examination of multiple parameters using a single class database of a single class of component’s parameters, such as proteins, pesticides, sugars for identification.
14. Non-targeted analysis is important in detecting the presence of non-intentionally added substances (NIAS) in food contact materials, especially in recycled materials. The importance of input materials to the recycling process and what may then be present was stressed in the interim COT position paper on ocean bound plastic. A second reason is the detection of proteins, in respect with novel proteins (for example from insects), these can cause an added challenge with regard to unknown allergens, due to currently unknown substances in novel protein products. This adds additional complexity beyond the known issue of unintended cross contamination with allergens in products and needs to be addressed through risk analysis of the supply chains.
15. The future and next steps were outlined. Literature must be collated with regard to reviews on the definitions and nomenclature, method performance and the quality of results including what reference materials were available.
16. There will be a reliance on compilation of big data sets. This may include considering how artificial intelligence can best be used to compile data in a fair, accountable and transparent way.
17. The ultimate goal is agreed validation procedures covering a range of concentrations and discoverable compounds of interest. An interim goal is to collaboratively test these methods. This is currently a voluntary exercise in the JEG but will eventually require increased effort and potentially external help in regard to funding.
18. The final topic in this presentation was NAMs. Public opinion is a key issue in the development and use of NAMs. The public are likely to be risk averse in the face of new regulatory methods and distrust what is perceived to be new and untried. It may be seen that NAMs are being forced due to changes in cultural attitudes rather than being innovated as a solution to a tangible problem and a convenient improvement made possible by modern developments. It will be important to engage with consumers and citizens. A suggestion of discussing NAMs at the upcoming FSA Board and Business Committee meetings, for example, would be a key way to accomplish this engagement.
19. However, scientific divergence should be avoided where possible, with a focus on a pool of expert and local authority activity being needed.
Dr Camilla Alexander-White (Royal Society of Chemistry Policy) then presented on chemical policy over the next 5 years and the bigger picture in general.
20. Dr Camilla Alexander-White described various recently published strategies including the FSA strategy and the HSE Strategy, and stated that the Department for Environment Food and Rural Affairs (Defra) are developing a new 20 year strategy on chemicals in addition to Defra’s Environmental Strategy 25 year plan (2018). Within the Environmental Strategy, Chapter 4 sets out how to increase resource efficiency and reduce pollution and waste including publishing a Chemicals Strategy.
HM Government’s Reforming the Framework for Better Regulation was also briefly mentioned.
21. The Royal Society of Chemistry (RSC) has been thinking what a chemical strategy should look like and its drivers and scope. The drivers to develop a new
UK chemicals framework that is fit for the future of the UK and international trade ambitions are: economic prosperity, wellbeing and quality of life improvements for citizens and wildlife. This is related to ‘trust in chemicals’ for the purposes of business and trade. The strategy should build trust, economic prosperity, wellbeing and quality of life through four pillars: education, innovation, circular economy and regulation.
22. Dr Alexander-White also introduced the recently published RSC risk based policy position on EDCs and discussed how Endocrine Disrupting Chemicals (EDCs) are best regulated in the context of promoting globally harmonised regulation, informed by collaborative science and research evidence. It announced that globally, there is a new United Nations Panel on Chemicals Waste and Pollution.
23. Questions raised included: are additional animal testing or exposure assessments required? For example, for Perfluoroalkyl and Polyfluoroalkyl Substances (PFASs), chemical regulations such as REACH may not be sufficient as the sole regulatory instrument to ensure proper management. However, would a NAMs-based approach be more appropriate? The RSC has published A proposed framework for risk-based PFAS regulation.
24. New ways of regulating the chemicals sector were then discussed including a response from the RSC to the House of Commons Environmental Audit Committee. It was stated that “the UK needs a clear, simple and enforceable regulatory framework relating to chemicals that balances the needs of research, innovation and trade with protecting citizens, wildlife and the environment. Regulation aims to ensure continued environmental protection from hazardous chemicals that can have harmful effects. Regulation must also enable industry to innovate and develop new products, using existing and new chemicals as raw materials, all of which can be traded internationally.” The RSC response to the house of Lords Committee post EU exit, on regulation of products, stated that “if divergence is to be considered and implemented, the RSC call on government to:
a) maintain harmonisation of the evidence-base between the UK and the EU.
b) put chemical safety science at the heart of regulatory decision-making.
c) look at regulation in the context of a longer-term UK chemical strategy.
d) make regulation decisions based on a set of defined and transparent principles.
e) ensure decision-making frameworks are transparent and have clear accountability.”
25. Areas should be identified where the UK could be a world leader e.g., NAMs and Next Generation Risk Assessment (NGRA) as applied in food and chemical regulations. This will ensure that leading scientists are engaged in UK and international science-policy interfaces.
26. Finally, there were three key asks of government in order to build confidence in UK Chemicals globally through regulation and science by: establishing a UK Chemicals Agency as a branded beacon of regulatory excellence worldwide; establishing an Applied Research Institute as a beacon of scientific excellence worldwide to directly support the Agency funded by government, but independent; and ensure transparency of decision making on chemicals safety and provision of information to workers and consumers.
Session 1: The landscape of regulation post EU exit
27. The first of three sessions looked at the landscape of regulation post EU exit from various perspectives including Chief Scientists, industry, academics and Scientific Advisory Committees.
Professor Robin May (FSA Chief Scientific Advisor) presented on opportunities and outlook for UK food regulation post EU exit.
28. Professor Robin May introduced the challenges of EU exit concerning international collaboration, data sharing and the flow of expertise in and out of the UK. It was stated that there is a need to identify those opportunities to maximise their potential, going forward. Two areas were then highlighted:
29. Regulatory framework: The role of innovation in the food sector such as alternative proteins, insect proteins and 3D printed food. Specifically, the FSA focus on gene editing and the Genetic Technology (Precision Breeding) Bill. This Bill is to make provision about the release and marketing of, and risk assessments relating to, precision bred plants and animals, and the marketing of food and feed produced from such plants and animals; and for connected purposes ( UK GOV Genetic Bill).
30. Methodologies: Computational modelling and artificial intelligence are great opportunities in predicting toxicology. It was stated we should invest further in computational modelling for the purpose of toxicological assessment.
31. Finally, it was stated that there is a need to keep up with scientific developments and sharing these will be key for the future.
Professor Isabel Oliver (UK HSA Chief Scientific Officer) presented an overview of the UKHSA role.
32. The role of the UKHSA, an executive agency of the Department of Health and Social Care (DHSC), is to detect, prevent and control threats to human health. Their role is to protect the public from communicable diseases and health effects from exposure to hazards including those of a chemical, radiological and a biological nature.
33. As a science-based organisation, the scientific evidence is developed and applied, to arrive at health outcomes. This involves scientists working in partnership with academia, across government departments and other organisations. An example of this is in the assessment of chemicals, where partnerships with departments including the FSA, the Health and Safety Executive (HSE), and Defra take place to ensure health threats are responded to effectively.
34. It was highlighted that the UK HSA have toxicologists working across government and internationally to work to protect the public from hazards including harmful effects from chemicals, by providing impartial, evidence-based advice. The UK HSA are currently developing their first science strategy, which will cover their vision and ambition for environmental public health. At the UK HSA, it is recognised that health is determined by environmental factors alongside other factors, including hazards of human and natural origin.
35. The UK HSA highlighted their aims to strengthen their scientific capabilities, to increase the impact of their work on health outcomes and the prosperity of the country. Their aim is to strengthen the scientific evidence base to inform appropriate standards and regulations for potential hazards including radiation, chemicals and biohazards, with an aim to build public confidence that these have been addressed and are well regulated.
36. The work of the UK HSA is primarily on hazard characterisation, exposure and risk assessment for chemicals, biologicals and radiation, which includes research into the human health effects of exposure to chemicals in humans. This allows UK HSA to advise partners and government. They also undertake biomonitoring to determine human exposure to natural and man-made chemicals in order to fully evaluate the risk. The UK HSA emphasised their commitment to reducing exposure to hazardous chemicals in food in order to prevent ill health of the public.
37. There are challenges, including chemicals entering the market, whose impact on human health is not fully understood. In future, the UK HSA plans to invest resources in this area and to strengthen collaborations with academia and other organisations.
Dr Andrew Smith (Head of Unit for REACH and CLP Delivery, and for specialists in toxicology and ecotoxicology in the Chemicals Regulatory Division of the Health and Safety Executive (HSE) presented on regulatory perspectives post EU Exit.
38. The UK leaving the EU has led to possible implications such as data loss, loss of technical support networks, peer review and project management. However, it was acknowledged that, there are some opportunities that have arisen from EU exit. There is an opportunity for the UK to deliver chemicals regulation effectively in an environment separate from that of the EU. It was stated that HSE are not looking to replicate the current EU approach but instead will be working with the Environment, Food Standards and Health Security Agencies, and others across the UK, to deliver its regulatory programmes for chemicals, biocides and pesticides. Independent scientific advisers have stated that currently the UK is in a good position to undertake this work, and there have been discussions as to how Defra and other UK bodies can engage with the Organisation for Economic Co-operation and Development (OECD), for example on test method and guideline development. It was emphasised that the HSE acts as the regulatory authority or “Agency” for chemicals biocides and pesticides. HSE’s Chemicals Regulation Division (CRD) is not a research institute. It must operate effectively and transparently, delivering regulatory outputs in a timely and cost-effective manner. CRD aims to protect both workers and the general public from chemicals, as well as the environment. Following EU-exit, digital solutions are being developed to help build capacity and improve the regulatory service provided for duty holders.
39. The challenges around building sufficient technical capability in CRD were highlighted. There has been a significant recruitment of new graduates and early career scientists, training now being a significant task. Looking ahead, new approaches will be needed to help ensure specialist staff are retained in sufficient numbers to enable CRD to operate as a stand-alone regulator, outside of the EU. There is also a need to recruit experienced specialists to train and support these new scientists. However, it was highlighted that there appears to be a shortage of skilled mid-career scientists available to recruit in the UK. Currently, HSE has several vacancies aimed at mid-career toxicologists required to address this capability issue.
40. Considering the REACH Regulation, HSE no longer has access to EU data and there is currently therefore no substantial UK database of chemicals of the kind maintained by the European Chemicals Agency (ECHA). There are also issues for industry duty holders, as they can no longer use EU data to seek registration or authorisation of their chemicals. HSE is currently working with Defra on getting better UK data. However, it could become more difficult to prioritise what chemicals justify regulatory intervention in the UK, as collaboration with EU Member States to gather and assess data is not possible. There will also be an additional cost to deliver this.
41. Regarding the Classification, Labelling and Packaging Regulation, it was explained that HSE are obligated to track progress with the harmonised hazard classification of substances in EU CLP, but there is scope for this British system to make its own decisions.
42. In both these areas, CRD is supported by scientists in the Environment Agency.
43. A key challenge with biocides and pesticides is the very significant volume of work mandated by the relevant regulatory frameworks. For the specialists in CRD, there are some significant regulatory issues to be worked through for both the active substances and the products being placed on the market. CRD covers both the environmental and human health risk assessments for biocides and pesticides. CRD is in the process of investing in a transformation programme to improve the digital tools used by its scientists to manage and assess technical data and enable a better service to be provided to duty holders.
Mr Charles Nancarrow (National Audit Office) substituted by Professor Tim Gant (Principal Toxicologist UKHSA) presented on National Audit Office Regulating after EU Exit Report.
44. The report considers three regulators whose work has been significantly affected by EU Exit. It draws out common issues and challenges to help inform regulators and policy departments as they develop regulation after EU Exit, both in the three areas covered, and more broadly across government. The report emphasises the government’s views that EU exit provides an opportunity to change food and chemicals regulation in the UK.
45. The report examined the HSE’s role in chemicals regulation, in particular the Chemicals Regulation Division; the FSA’s role in regulating food safety and standards; and the Competition and Markets Authority’s (CMA’s) roles in enforcing competition and consumer protection law; operating the Office for the Internal Market; and preparing to provide subsidy advice within the UK subsidy control regime.
46. The discussion considered that the EU Exit is an opportunity for change. It was emphasised though, that loss of access to a range of data sharing and cooperation arrangements is one of the challenges that will need to be addressed. The change needs to be underpinned by science, ensure competitive markets and reduce admin burdens.
47. To assist in the development of regulation and data development there is a base of good sound science across government. This needs to be used to monitor scientific developments and use the potential for divergence within devolved admins of the UK as an opportunity and provide training where appropriate.
48. However, the demand for authorisations in the UK is greater than originally thought, so there is a challenge for government finance and scientific resource, and for the further provision of these resources from the academic sector.
49. For these reasons there will be a need to look at how the landscape is being developed across the board including strategies, new methods, recruitment issues, capability building and the amount of investment required to take the regulations forward.
50. Recommendations of the report were as follows:
a) While government as a whole is still working on its future direction for regulation, regulators need to develop, with policy-makers, their long-term strategies and objectives.
b) Now there is more clarity on demand for new regulatory functions, regulators should review the plans they developed before EU Exit in the light of what they now know about their capacity and their workload.
c) Regulators should ensure that, as soon as they are able, they provide clarity to stakeholders on their intended plans and the timelines for any planned changes.
d) Government should draw on the findings in this report as it considers the future of regulation after EU Exit more widely – for example, in its work in response to the consultation on the framework for better regulation.
51. Ultimately, it was stated that the most important thing is for industry, academia and regulatory agencies to work together in order to review deadlines to match workload and resources.
Dr Dave Bench (Crop Sciences) presented on industry perspectives.
52. Dr Dave Bench introduced his background having worked on crop protection and digital data protection in HSE for 6 years to provide safe and robust regulation for the public and maintain high standards for foods.
53. Produce should be sufficiently safe, healthy and affordable whilst high standards of protection for human health and the environment are maintained.
54. Some suggested opportunities include support of innovation for post EU Exit through:
- a coherent agriculture policy (to include the protection of the interests of Northern Ireland) to encourage innovation and enable UK growers to compete globally;
- a science-based, enabling regulation could allow innovation to solve significant global challenges;
- a clear Government direction for regulators and better regulation and sensible use of regulatory autonomy.
55. Some of the numerous challenges since EU exit were then discussed. There has been a regulatory shift. For a new active substance approval it must go through the UK regulatory system. Bringing a product to the UK is key to expanding the market, but this involves huge direct and indirect costs for the companies.
56. The problems include insufficient capacity to support the market; the UK has less tools than EU; there are 3 different markets (the UK, the EU and Northern Ireland). It was noted that Northern Ireland is currently a different market.
Professor Alan Boobis (Chair of COT/Imperial College London) gave an academic/scientific advisory committee perspective, noting that he was providing personal views informed by being the Chair of the COT.
57. Currently, hazard characterisation is typically based on data from laboratory animal species, but this will change in the future to more reliance on in vitro and computational methods. SACs should be taking the lead on this. In considering what level of certainty is needed from a new method, it is important to recognise that all assessment methods are models, even clinical studies in small numbers of humans, and hence there is always the need for extrapolation when considering the whole population.
58. Health-based guidance values (HBGVs) are established by dividing a point of departure by uncertainty factors to allow for differences between species and within the human population. Exposures below the HBGV are considered to be of minimal risk and exposures above the HBGV may pose a potential risk. There is a tendency to treat HBGVs as set values, yet the application of conservative assumptions in setting HBGVs can lead to some very low HBGVs. In addition, a concept of non-threshold effects based only on the analysis of empirical data has arisen amongst some risk assessment scientists, including the SACs. Professor Boobis explained that it is also important to take the mechanisms into account. When considering key events and homeostatic processes, there must be limits in the dose-response distribution, below which no-one will be susceptible. The issue has partly arisen due to the use of probabilistic methods and the need to decide which percentile we accept as “safe”.
59. Professor Boobis then discussed risk assessment. The separation between risk assessment and risk management now needs to be rethought. It has been useful in restoring public confidence, but risk assessment is not an end in itself. It is an input into policy, and some risk assessments are not fit for this purpose if they use extremely conservative assumptions.
60. Risk mitigation is rarely a zero sum game. Zero-sum game is a mathematical representation in game theory and economic theory of a situation which involves two sides, where the result is an advantage for one side and an equivalent loss for the other. As an example, for electronic nicotine delivery systems (ENDS), the UK recognises that the alternative, conventional cigarettes, causes far greater harm, so it is not the absolute risk of ENDS themselves that is the primary consideration from a public health perspective, but the risk relative to smoking cigarettes. There needs to be consideration of establishing a framework where risk assessments are fit for purpose. The problem formulation needs to reflect the degree of conservatism required.
61. The issue of mixtures risk assessment was then addressed. Is it possible to agree upon a pragmatic approach to assess combined exposure to multiple chemicals without being overly conservative? Even for pesticides, where risk assessment of combined exposures is most widely implemented, there are large differences in the approach used in different regions; for example, the US Environmental Protection Agency (EPA) groups pesticides based on their similarity in structure and sharing a common mode of action, whereas the European Food Safety Authority (EFSA) groups them based on common adverse outcomes (i.e. adverse effects). The EPA uses adverse outcome pathways (AOPs) or modes of action to exclude pesticides from groups whereas EFSA uses AOPs only to include pesticides in groups.
62. Professor Boobis raised the topic of the microbiome, and that there is no agreed position on how to address risks to health from effects on the intestinal microbiome. For antibiotic veterinary medicines there is an agreed approach, aimed at protecting against disruption of the colonisation barrier and the development of resistance to the antibiotic, but increasingly it has been suggested that any substance that enters the gastrointestinal tract might affect the microbiome. The UK needs to consider a sensible approach to address this concern, which does not require testing of every chemical against all taxa in the microbiome.
63. In summary, chemical risk assessment has to be able to help address the challenges posed by serious threats to food security, such as climate change, wars and pandemics, and the speed of transition resulting from the green revolution, new energy sources and the combined threat to humans and the environment of biodiversity loss. Risk is not an absolute and we often need objective quantification of risks and of benefits, including indirect benefits such as food security and socioeconomics to address these issues.
64. The UK needs to develop a systematic approach to risk assessment of combined exposures to chemicals, and we need to develop a UK position on an appropriate strategy for assessing potential risks to the intestinal microbiome.
Dr John Doe (Liverpool John Moores University, Honorary Research Fellow) presented on chemical risk assessment and regulation.
65. Dr John Doe stated that the EU regulatory framework on chemicals, is based on high level scientific advice and application of the precautionary principle. It is considered to be one of the strictest, if not the strictest, in the world. It ensures a high level of protection of human health and the environment. The overall aim of all EU legislation on chemicals is to achieve a high level of protection of human health and the environment by minimising exposure to hazardous substances and by encouraging substitution of hazardous substances with less hazardous chemicals, as far as technically and practically possible. This remains the overarching objective of the UK.
66. There is an opportunity to make chemical risk assessment more ‘fit for purpose’ now the UK has left the EU.
67. One of the drawbacks of EU policy, is that it is focused on minimising exposure to hazardous substances (e.g., based on a classification criteria) rather than characterising the hazard in more detail.
68. One way the UK could change the risk assessment paradigm is by characterising these hazards in more detail. Dr John Doe then introduced the paper “The codification of hazard and its impact on the hazard versus risk controversy” (Doe et al., 2019) whilst explaining the three levels of hazard codification:
Level 1: Hazard identification presence or absence of a class of adverse effect. Yes or no binary choice (limited banding) method;
Level 2: Compartmentalization of hazard, first by nature of the adverse effect and then by potency by banding with several categories (typically 3-5). This is a semi-quantitative method.
Level 3: Description of the nature of an adverse effect and the derivation of a health-based guidance value (e.g., acceptable daily intake, reference dose, derived no effect level) to establish safe levels of exposure. This creates a continuous response based on quantitative dose response risk assessment.
69. The underlying reason for the controversy between hazard and risk is the use of level 1 hazard codification schemes in situations where there are ranges of severity and potency. Existing level 1 codification schemes should be reviewed and developed into level 2 schemes where appropriate.
70. In particular, moving away from ‘level 1’ hazard characterisation (binary approach) to a level 2 (compartmentalisation, quantitative) approach where appropriate. An example was used in the form of classification of carcinogens. A tiered approach was used to describe group 1 carcinogens looking at each chemical’s effect, dose response and mode of action (i.e., further characterisation).
71. From this the chemical can potentially be put into one of 3 categories:
- Primary/Direct effect i.e., directly mutagenic.
- Secondary effect through e.g., hormone response.
- Tertiary or collateral effect e.g., cell repair and proliferation after a cytotoxic effect.
72. Following this, chemicals could be classified into one of 3 categories of potency: low, medium or high. The potency vs effect matrix can then be used to characterise the hazard.
73. The current EU REACH mandated approach was then analysed; this is where a set of studies and data must be produced based upon the annual tonnage of production of the chemical. The disadvantage with this is that it is so prescriptive it can stifle innovation and discourages the use of NAMs.
74. Some forward looking thoughts were then presented. Classification should be based on severity and potency. Care should be taken in mandating downstream risk management based on classification. Legislation should demand scientifically valid answers to health concerns but not mandate the methodology. Methodology should be specified in guidance which can be revised as the science and methodology advances. The Scientific Committees can advise on the science and methodology.
Cross cutting themes of landscapes discussions
The following questions were used as prompts for the discussion:
• Is there a need for any change in the UK approach to regulation of food and chemicals from that inherited from the EU?
• Would a differentiated approach to food and chemicals regulation between the EU and the UK be sustainable politically and with the general public?
• Would a differentiated approach bring real benefits to industry and the public, or would it create complexity and greater cost given the relatively small size of the UK market?
Roundtable Discussions
Themes
75. Standards: It was stated that there needs to be a good balance of opportunities that may arise from a UK approach and ensure standards are maintained as well as possibly even improved. It was agreed that the UK should not lower its standards in regulation.
76. Public Engagement: Public confidence was then discussed. The public need to be fully informed on what has been decided and why to ensure that trust in food and chemical safety is maintained. This will require increased transparency with the public to affirm that change in regulation is not a decrease in safety.
77. Risk Communication: Risk communication can influence the public’s perception, therefore engaging with the public in the right way is key. It will need to be made clear that the UK government understand their concerns and explain that some issues may have uncertainties and associated implications.
78. NAMs: The UK need to identify new methods for testing and assessment, to help reduce data gaps and to improve human relevance. The way to increase confidence in NAMs is to show that they are reliable, but above all else that they are sufficiently protective of human health and the environment. However, lack of funding makes this difficult to confirm this. It is important to explain the value of using NAMs in food and chemical risk assessment. The public should be made aware of the value of NAMs and that they are not solely for the benefit of animal replacement benefit but also to improve the science overall, providing more reliable and relevant assessments. Public acceptance on the use of NAMs will be fundamental in the progress of their acceptance. It will also need a public perception paradigm shift, not just a scientific one. Funding was brought up as a barrier, as it is very limited and few, to no resources are currently going into the validation of NAMs. Moving forward, the UK should be expanding horizons and learning from other regulators.
79. Biomonitoring: It was raised that as biomonitoring becomes more prominent, there might be a demand from the public to reduce exposure to chemicals.
Session 2: International and Divergence
80. The second of the three sessions looked at international toxicology projects, the OECD role in chemical regulation, government projects and possible new opportunities in divergence.
Dr Fatima Nasser (Department for Environment, Food and Rural Affairs)
gave a talk on the importance of engaging with the Organisation for Economic Co-operation and Development (OECD) for UK chemical regulation.
81. Dr Fatima Nasser provided background on the OECD and how the work of the OECD supports UK objectives. This includes evidence-based policy making, developing international standards and provides a forum for open discussions. The OECD also work on international mutual acceptance of data (MAD).
82. The UK vision would be to collaborate internationally to both shape and deliver policy goals on chemicals and waste through the development of analyses, tools, standards and guidance.
83. This will not only provide the opportunity to collaborate and lead internationally but for the UK to have its own chemical regulation. There would be the opportunity to think around chemical testing methods to demonstrate chemical safety. It would allow the UK to design its own programme and shape its policies. This would promote the UK’s own scientific priorities and allow scientists to input into international guidance. Scientists and regulators could participate as part of UK delegations.
84. Dr Nasser then explained the OECD’s Environment Directorate and the Environment, Health and Safety (EHS) Programme which provides policy tools and evaluations focused on environmental reviews, indicators and outlooks; climate change, biodiversity, water and waste; decoupling environmental pressures from economic growth; green growth and chemical safety and biosafety. The OECD Environment Directorate helps countries design environmental policies that are both economically efficient and effective at achieving their environmental objectives. Finally, it was discussed that it will be important to engage with the OECD EHS programme, as the UK expects to remain an influential player within this forum post EU-exit.
85. The OECD provides a valuable opportunity for the UK to continue to exhibit international leadership. Engagement with the OECD ensures continued visibility post EU exit and guarantees that the UK will still be perceived as a global and open economy. The EHS programme has objectives that parallel those of the UK for managing the use of chemicals to protect environmental health. These coincide with Defra’s objectives of the 25 Year Environment Plan. Harmonization through the MAD system should eliminate repetition of work and ensure that tests follow the OECD Test Guideline of Good Laboratory Practice which will lead to financial benefits. The tools and policy guidance, into which the UK contributes, and relates to the sound management of chemicals, are available to all countries giving the UK international influence. Therefore, the OECD provides an open forum where scientists and regulators can work together and share technical and scientific information on chemical safety which influences policy.
Dr Hannah Littler (Defra) presented on the role and importance of international organisations and collaboration with a new direction for policy and supporting evidence.
86. Dr Hannah Littler introduced the opportunities and challenges of EU exit. There are numerous opportunities for the UK, such as being able to make its own regulatory frameworks according to the best available evidence. This will allow the UK to make its own decisions about food and chemicals regulation (the aim is to take ideas from both inside and outside the EU, and act on the best available evidence). It also means the UK can continue to build its reputation as a world leader in these matters. However, Dr Littler acknowledged that regulation of food and chemicals is a global problem, which requires international collaboration. Therefore, the UK must remain a member of the international regulatory community. The UK is free to make its own decisions about domestic Food and Chemicals Regulation. Could the UK be a leader on novel strategies for chemicals management? e.g. NAMs, risk-based chemical assessment etc.
87. Dr Littler then stated the role of the OECD, as an example of an international organisation which the UK can still collaborate with. The OECD covers not just the EU but also the US (United States), Japan, and Canada. Another example for international collaboration is the UN (United Nations), which currently has several programs in respect of chemicals regulation, such as the UN Environment Program (UNEP), and the strategic approach to international chemicals management (SAICM). Some other organisations were also discussed, such as the World Health Organisation (WHO).
88. International collaboration is vital to position the UK as an active and open member of the international regulatory community and a leader in chemicals management. The UK should engage with international fora to: demonstrate UK scientific and regulatory capability/leadership; share and gather evidence and knowledge to support domestic regulatory policy; steer international chemical regulations/research in a direction that benefits the UK as well as international partners.
89. Overall, EU exit presents a new direction, which the UK aims to use to its advantage, to strengthen its position as a more independent leader in the international food and chemicals regulation space.
Ms Melanie Foster (Defra) talked about Defra’s Chemical Strategy.
90. Ms Melanie Foster introduced the context for the UK’s chemical strategy. In 2018, the Government published its 25 Year Environmental Plan with an overall vision to “To restore and enhance the environment for the next generation by making our air purer, our water cleaner, our land greener and our food more sustainable.”
91. The Environment Act (2021) made the 25 Year Environment Plan the first Environment Improvement Plan which is required to be updated every 5 years. An updated Environmental Improvement Plan will be published in January next year. It will include a chapter on the chemicals goal of ‘Managing Exposure to Chemicals’, setting out the plan of work on Chemicals for the next 5 years. In particular, Goal 9 was discussed, which states that: “we will make sure that chemicals are safely used and managed, and that the levels of harmful chemicals entering the environment are significantly reduced” (25 Year Environment Plan UK GOV).
92. The development of a new Chemicals Strategy will provide an opportunity to set out future priorities, and to articulate a framework for how we intend to approach chemical regulation now that the UK has left the EU.
93. Leaving the EU has changed the context of the UK’s work on chemicals management and UK priorities. There is a need to continue to effectively operate and develop legislative functions, repatriated from the EU. The UK also needs to develop policy to tackle emerging chemical threats but that also supports the UK economy and protects human health and the environment. The UK policy will also need to promote the interests of the UK chemicals sector internationally and reinforce commitments to strengthen the global environment and health protection.
94. At the end of the transition period the idea was to replace the work of European Chemical Agency (ECHA) and continue to deliver and promote the Chemical Strategy.
95. The key themes of the Chemical Strategy were then presented.
- The ‘Driving innovation and sustainable chemistry’ theme will cover ways to support the transition to and mainstreaming of sustainable chemistry, including safer alternatives.
- The ‘Tackling priority and future chemical risks’ theme will look at early warning systems and horizon scanning techniques to identify, manage and enable early action to minimise the threat of emerging chemical risks.
- The ‘Managing Chemicals in waste’ theme will cover managing the risks posed by chemicals at end of life and taking action to reduce future legacy problems to support the circular economy e.g. ensuring we track hazardous chemicals in products.
- The ‘Regulatory framework’ theme will use effective, evidence-based and proportionate regulation of chemicals to meet the priorities set out under the other themes, including: how the UK can evolve its regulatory framework now that the UK is no longer part of the EU, to ensure improved domestic outcomes and how mechanisms can be developed to address key chemical policy issues more effectively. This includes ‘grouping’ approaches and consideration of ‘essential use’ as a tool/concept for risk management. Effective regulation is the starting point for ensuring the benefits of chemicals are maximised and the risks that they pose are safely managed. The UK needs to ensure high levels of protection for human health and the environment through effective, efficient, and proportionate chemicals regulation.
- The final theme presented was ‘International work’. This will include promotion of the UK as a leader in science (e.g., sustainable chemistry); opportunities to show international leadership that is backed by domestic excellence; influence and collaborate with other nations to facilitate trade; and recover biodiversity as well as protect human health.
96. Ms Foster then discussed the next steps which include stakeholder engagement through workshops and timelines in the Chemical Strategy. Ambitions for the UK will need to be established, including a goal to strive to eliminate animal testing through the embedding of NAMs in the UK, and working internationally to drive the embedding of NAMs at a global level as well as ensuring that there is an emphasis on data sharing. Another ambition is to develop policy options for unintentional mixtures under UK REACH.
97. Finally, international alignment and collaboration is key. The UK is already contributing through OECD and through the EU research programme ‘Partnership for the Assessment of Risk from Chemicals’ (PARC).
Dr Ovnair Sepai (UK HSA) presented on ‘Partnership for the Assessment of Risk from Chemicals’ (PARC).
98. In 2018, ANSES (the French Agency for Food, Environmental and Occupational Health & Safety) presented a proposal to Horizon Europe to look at setting up an EU toxicology programme to develop next-generation chemical risk assessment. It is a co-funded initiative involving 28 countries of 200 organisations in a 7-year partnership. The UK is a partner but there are issues and some implications with funding.
99. The programme is a platform for applying and developing advanced tools. The current gaps in knowledge include assessment of chemical mixtures, method development, use of NAMs and regulatory acceptance of NAMs.
100. There are several work packages which include: findability, accessibility, interoperability, and reusability (FAIR) data, innovation in regulatory risk assessment, a common science-policy agenda, safe and sustainable, hazard assessment, building infrastructural/human capabilities, monitoring and exposure. The monitoring and exposure work package builds on the human biomonitoring programme of work. Hazard assessment should move to next generation risk assessment with moves to incorporate in silico and in vitro studies. These new methods will also need to be validated. The work plan on risk assessment has a lot of overlap with programmes on hazard and risk assessment, mixtures, and real-life exposure.
101. There is a need to develop integrated approaches such as the Integrated Approaches to Testing and Assessment (IATA) and to engage with non-government organisation (NGOs), industry and the public. It is possible for the UK to input data into every aspect of PARC due to the UK representation, and mirror some of the ambitions in the EU. There needs to be engagement at the European level and links with the UK Chemical Strategy.
102. The vision is to protect human health and the environment; contribute to a non-toxic environment and a circular economy; generating the best science to answer regulatory questions and ensure that science meets regulatory needs.
Ms Laura Holden (University of Birmingham) presented on (Precision Tox).
103. Precision Tox is a Horizon 2020 project. The goal of Precision Tox is to improve chemical safety assessment to better protect human health and the environment by using non-traditional test species, multiple fields of knowledge, and powerful computational approaches to understand which chemicals are toxic and why.
104. The aims of the Precision Tox programme align with the aims of UK REACH; high level of protection of human health and the environment and include the registration, evaluation, authorisation and restriction of chemicals.
105. Currently in EU REACH, information requirements increase based on the tonnage produced. This can lead to chemicals, where their effects are less well characterised, being on the market. REACH aims for the reduction, refinement and replacement (3Rs) of animal studies. In the current system, industry provides the information, and the regulator evaluates it. Ms Holden noted that there are many imported and distributed chemicals for which there is very little information. Precision medicine was briefly discussed. It was stated that omics technologies and biomarkers have billions in global spend and that these approaches could be used for identifying potentially hazardous chemicals.
106. Comparative toxicology was then discussed. This focuses on 3R compliant model species which includes invertebrates such as water fleas, among others. This is based on the principle that toxicity pathways can be conserved through evolution. It is also possible to observe effects in such whole organism models and populations. This is due to shorter lifecycles and large populations.
107. 3R compliance can be achieved by the use of multiple models in a high throughput, tiered approach. This would allow understanding of variation in susceptibility.
108. By improving the regulatory approach to chemicals, it would be possible to eliminate the need for observation of apical endpoints through a greater level of knowledge of a chemical’s mechanism of action.
109. A progressive precision regulatory framework was then presented which included biomarker levels and criteria including: regulatory and scientific relevance at each level; reliability; transparency; access to data.
110. Finally, the redirection of toxicity testing and regulation was discussed which included the topics of a NAM Toolbox of biomarkers, protocols, reporting templates, training, guidance; 3Rs and a framework encompassing human health & environmental hazard information; precautionary principle; regulatory efficiencies post EU Exit and the biomarker market.
Dr Miriam Jacobs (UKHSA) presented on the project: GOLIATH (Generation of NoveL, Integrated and Internationally Harmonised Approaches for Testing Metabolism Disrupting Compounds)
111. GOLIATH is an EU funded project from Horizon 2020, with an aim to develop testing approaches for metabolic disruption, which is part of the Eurion cluster of projects.
112. GOLIATH objectives aim to improve the understanding of endocrine modes of action (MoA) of metabolic disrupting chemicals (MDCs); develop assay candidates based on confirmed MoA and key biological effects in target tissues relevant for adversity; (pre)-validation on in vitro test methods; to develop in vitro assay candidates into (pre-)validated test methods within OECD; augment the chemical applicability domain of the CYP induction HepaRG test method and regulatory applications; develop a conceptual IATA for MDCs.
113. MDCs comprise ‘Any Endocrine Disrupting Chemicals that alters susceptibility to metabolic disorders (e.g. obesity, diabetes, non-alcoholic fatty liver disease)’ which includes: class of EDCs that affect energy homeostasis; affect multiple endocrine mechanisms and cell types implicated in metabolic control; affect gene expression and biosynthesis of key enzymes, hormones and adipokines essential for controlling energy homeostasis.
114. The main key exploitable results of the GOLIATH project are: in silico open access tools for predicting molecular initiating events; AOPs/mechanistic evidence for metabolic disruption; IATA for MDCs; and international input, harmonisation, and acceptance.
Roundtable Discussions
The following questions were used as prompts for the discussion:
- What is the relevance and importance to the UK of international involvement and contribution to international bodies?
- How do we ensure involvement from academia/government and industry into research projects and knowledge translation to strategies and international guidance and protocols?
- How to we develop and translate new research and knowledge into regulation and how is this supported and applied in a tripartite manner (industry, academia, government)?
Roundtable Discussions Themes
115. Delivery: The pace of scientific development and outcome were discussed. The OECD relies on consensus, so test guidelines, IATAs etc take time to be agreed. Also, research needs to deliver concrete outcomes, e.g. EU research on alternatives to animals, which influences programme design. The work done at the OECD generates tools and standards that are agreed upon by all participants, but it is the policy decisions that determine how we use those tools. The time input into generating those tools helps to avoid duplicating effort and allow for the mutual acceptance of data, therefore gaining large benefits.
116. Communication: Communication was discussed. The group suggested that there should be more communication among agencies and academia as well as scientists and risk assessors.
117. Biomonitoring data: It was asked whether there will be UK biomonitoring data to submit into the European PARC. The UK will participate and samples are now being collected from the Health Survey for England for biomonitoring. Other further UK data will also be collected within the PARC project. The Environment Agency collects environmental data and there are also food surveillance data too. It was emphasised that being able to share UK data is really important for collaboration.
118. NAMs: There were discussions on how to use NAMs in regulation. EFSA discussions on NAMs have indicated progress is very slow yet a number of suitable tools have been presented here. It was highlighted that HSE has freedom in some aspects of regulation and it was suggested that it should apply this approach to look for data and opportunities to utilise NAMs. However, it was noted that HSE has raised a call for evidence on tattoo inks and there is nothing stopping the scientific community providing data using new techniques. It would then be HSE’s challenge on how to use these data. HSE could seek advice from the COT, COC and/or COM. HSE are very open to using weight of evidence and taking different issues to scientific advisory committees for advice. The view was expressed that all available evidence should be integrated. Currently in risk assessments much of the available evidence is discarded. There has been some frustration with the lack of regulatory impact of EU programmes, despite the fact that many novel methods have been developed; NAMs tools are already available and are there to be used. The aspects that are missing are the validation and regulatory agencies integrating them i.e. development to implementation. The UK could put in a lot more effort here. EU collaborative programmes are a good way to obtain research funding. An excellent piece of work championing this is the ongoing FSA and COT NAMs roadmap, which will hopefully encourage acceptance and integration.
119. Methodologies: The International Programme on Chemical Safety (IPCS) was then mentioned. Early on, this programme was very successful in developing international risk assessment methodologies, e.g. MOA. Now there are few governments directly supporting this type of work at the IPCS. However, a new panel, the United Nations Panel on Chemicals Waste and Pollution, has been established with terms of reference similar to but much broader than those of the IPCS. Hopefully this new programme will maintain its funding and will be able to deliver.
120. Regulation and Policy: There is often a gap between policy and regulation. Despite Government involvement and clear regulatory guidelines the question of what level of protection should be delivered is never asked. Unless there is a link to that policy question there is just a proliferation of data requirements and guidance. The UK should be trying to find what is needed to take good quality decisions. An important aspect will be to ask what level of protection the UK wants to implement. Otherwise, it was highlighted that all of the studies with available data are unusable or not designed for the purpose.
Session 3: Major drivers for change and potential impact on chemical regulation & Indirect Effects: food prices, food security, supply chain, fraud | Food regulation/human health
121. The third and final session looked at food regulation and health through government strategies, drivers and effects, personal perspectives and CODEX Alimentarius.
Professor Rick Mumford (Deputy Chief Scientific Advisor & Deputy Director of Science, FSA) presented on FSA Strategy & Science: perspectives on future challenges.
122. Professor Rick Mumford introduced the FSA Strategy 2022-27. The FSA mission is food you can trust. FSA want to ensure that: Food is safe; Food is what it says it is and Food is healthier and more sustainable. Then some of the challenges faced from a risk assessors prospective were discussed. As per the seven guiding principles, the FSA remains science and evidence led.
123. The main roles played by the FSA were highlighted, including policy making, risk management decision making and risk assessment.
124. In terms of building the FSA’s science and evidence capacity, focus is on building science capability with the Science, Evidence and Research Division growing from 60 to over 150 people and including analysts, by the end of the recruitment period. This includes 90 staff in risk assessment.
125. The FSA also has the Chief Scientific Advisor, Robin May and the Science Council who deliver strategic advice and assurance as well as the SACs, who are essential in how the FSA delivers science and evidence. Access to national reference laboratories is also valuable in advising the FSA on areas such as new methods of analysis. Access to the register of specialists plays an important role in providing access to expertise from over 300 specialists.
126. The FSA also invest around 10 million pounds per year into their science, evidence and research programme, which focuses on specific areas of interest with a view to gain more scientific evidence and research.
127. Professor Mumford also discussed the FSA’s role in responding to emergencies, highlighting the work carried out surrounding Covid-19 and the Ukraine conflict. These included risk assessments carried out on the transmission of Sars-Cov-2 and the impact of the conflict affecting import of oils from the Ukraine and the issues arising from these emergencies, for example to the global supply chain.
128. Regarding food insecurity and its impact on food safety behaviours, evidence gathering allows the FSA to determine consumers views on food insecurity and how this leads to more risk-taking behaviours, such as ignoring use by dates. In addition to the drivers of change, some of the challenges were presented. These included: antimicrobial resistance, plastic pollution and climate change.
129. The implication of the changing food system and food safety was touched upon; particularly in terms of areas such as the future of meat, which has been the subject of recent discussions. Horizon scanning and looking at potential sources of novel protein and the implications for food safety has also been taking place.
130. The subject of driving innovation and new technologies was highlighted, and the policy and methodologies behind their regulation was raised as these present both new opportunities and challenges. This includes areas such as the use of genomics, which can be employed in the detection of food-borne diseases as well as the potential of NAMs. Engagement with innovators, and the role of the FSA in the area of regulatory science and how the FSA fits into the system was also discussed.
131. The inaugural food standards report published by the FSA and FSS was highlighted as an example of published data on the changing habits of consumers. This feeds into the area of regulated products. In the interest of openness and transparency, the FSA will continue to publish ongoing work.
132. The unintended consequences of replacement, and the implications, for example in the potential for microbiological contamination in the use of single use plastics, were discussed. The wider question surrounding risk appetite was an area highlighted as needing further discussion.
Mr Steve Wearne (CODEX) presented on Codex perspective on drivers for change and potential impacts on chemical regulation.
133. Mr Steve Wearne presented a talk on the Codex perspective on drivers for change and potential impacts on chemical regulation. Mr Steve Wearne is Chairperson of the Codex Alimentarius Commission and is currently on secondment from the UK FSA.
134. The Codex Alimentarius is a food code that is comprised of a series of general and specific food safety standards, guidelines and codes of practice that have been formulated with the objective of protecting consumer health and ensuring fair practices in the food trade.
135. The vision for Codex Alimentarius, set out in its Strategic Plan for 2020-2025, is to be where the world comes together to create food safety and quality standards to protect everyone everywhere. One of its strategic goals is to address current, emerging and critical issues in a timely manner. The food code currently includes 79 guidelines, 113 maximum levels for contaminants in food (covering 18 contaminants), 4,596 maximum levels (covering 376 food additives or groups of food additives), 232 standards (11 general standards, 221 commodity standards), 632 maximum residue limits for residues of veterinary drugs in food (79 veterinary drugs, Risk Management Recommendations (RMRs) for 13 veterinary drugs and 5,663 maximum residue limits (63 extraneous maximum residue limits (EMRLs) for pesticides residues covering 231 pesticides. It was emphasised that there is a continuing need for transparency in risk management to maintain trust, prevent misrepresentation of the science, and build a mutual understanding.
136. The UN Food Systems Summit in 2021 aimed to launch bold new actions, solutions and strategies to deliver progress on all 17 Sustainable Development Goals (SDGs), each of which relies on healthier, more sustainable and more equitable food systems. The Summit concluded that the global community must work together to transform the way the world produces, consumes and thinks about food.
137. On the 21st February 2022, the EU Council concluded that the EU has a strong willingness to explore, together with its partners, all the pragmatic ways of integrating sustainability considerations into the work of the Codex Alimentarius Commission. In June 2022, the World Trade Organisation published a Sanitary and Phytosanitary (SPS) Declaration for the 12th Ministerial Conference which aimed to respond to modern SPS challenges. It was concluded that the SPS Committee should explore how the implementation and application of the SPS Agreement can support the facilitation of global food security and more sustainable food systems. This should be done through sustainable growth and innovation in agricultural production and international trade International standards, guidelines, and recommendations developed by the Codex Alimentarius Commission, the World Organisation for Animal Health and the International Plant Protection Convention as the basis of harmonized SPS measures to protect human, animal or plant life or health should also be used.
138. In summary, the first of three drivers for change is the continuing need for transparency in risk management, to maintain trust, prevent misrepresentation of science, and build mutual understanding. The second driver for change is having harmonised trade measures and standards that facilitate the emergence of more sustainable food systems. The final driver for change is recognising how responses to some Covid challenges now provide us with opportunities to improve developments post pandemic.
Dr Izaak Fryer-Kanssen (FSA) presented on UK Regulation of Chemicals in Food Post-EU Exit.
139. Dr Izaak Fryer-Kanssen introduced the different and wide range of sources of contaminants and residues that can be found in food. These can enter at different stages in food production and can include primary production, in the growth of the food and secondary production, storage and transport.
140. These contaminants have varied risks and impacts on consumers. They may have acute risks, associated with a single exposure or exposure over a short period (e.g. hydrogen cyanide in raw bitter apricot kernels). There may be chronic risks, where harm arises through repeated, long-term exposure or the accumulation of the contaminant in the body, for example carcinogenic contaminants, or persistent organic pollutants which can pose a bioaccumulation risk. These risks can often not be fully eliminated. It is attempted to keep levels as low as reasonably achievable (ALARA).
141. Upon EU-exit, EU law became retained law (REUL) within the UK. In the future the FSA will advise ministers on new maximum levels MLs or changes to REUL. However, changing MLs is not always the answer to risk management issues.
142. The risk analysis process for the FSA was presented.
143. Dr Fryer-Kanssen presented a diagram showing potential risk management options besides altering maximum levels. This diagram posed a number of questions in a structured approach to risk management. These included: ‘Are exposures within tolerable levels?’ ‘If not, can the risk be managed by consumer advice?’ ‘For example, through effective communication?’ The next question posed was ‘can contamination be reduced at the source by production or manufacturing?’ Examples of this were teas and herbs in addition to soft drinks and acrylamide. The next stage is to assess whether there are alternative legislative approaches or decide that the current guidance is appropriate.
144. Finally, at the last stage it may be necessary to establish proportionate achievable maximum levels.
Professor Maged Younes (COT) presented on Food and Chemical Safety – Quo Vadis? Some Personal Thoughts.
145. Professor Maged Younes presented on some personal thoughts based on what he had observed over the years. There is often very little cooperation in science and lots of duplication. Competition for funding is sometimes unhealthy. There is hardly any incentive for applied or regulatory research. There is a publication bias, with negative results not being published and impact factors not favouring applied and regulatory research. There is little focus on risk science. If we cannot agree, then how do we communicate risks to the public? There is a lack of integration, e.g., of toxicology and epidemiology. There is fragmented scientific advice, with the potential for different approaches using the same data, and conflicts between the conclusions of different committees.
146. Internationally, there is a lot of duplication of effort which is a risk for the UK, post EU exit. There is political involvement in what risk bodies do. There is a lack of harmonisation and mutual acceptance of data outside of OECD countries. Standards, harmonised approaches and understanding would help with this. There is also a tendency for risk managers to tweak the science to suit their needs.
147. Prioritisation of applied research and risk sciences is needed, including the validation of NAMs: the promotion of collaboration across research; provision of sufficient funding; enhanced international research collaboration; and space to publish negative results. For scientific advice: cross-sector committees are needed, possibly including environmental health; structured approaches which allow for multidisciplinary input; continuous dialogue between risk assessors and risk managers; and harmonised frameworks and methodologies.
148. At the international level there is a need for harmonised methodologies, early exchanges and collaboration, better use of international fora, exchange between risk managers at the international level, and trust and mutual understanding.
149. Professor Younes outlined what he hoped to see from the UK in the future. This included promoting research on risk assessment methodologies, NAMs, big data and others, including validation of NAMs for regulatory purposes; dedicated funding for applied sciences; the development of further approaches to risk communication; and promoting international collaboration in risk science. Regarding scientific advice, the speaker hoped to see working in a more integrated manner, e.g., through joint committees; the integration of NAMs and big data into scientific advice; the avoidance of duplication through prior consultation; the promoting of joint assessments; and communicating in a clear and transparent manner. Internationally, we would hope to see the UK maintaining collaboration at the European level; promoting collaboration at the global level; enhancing the risk assessment capacities of developing countries; playing a major role in international fora; and acting as an independent entity in international disagreements in risk assessment opinions.
Dr David Gott (Head of Toxicology, FSA) presented on Food and Chemical Safety - personal thoughts over the years.
150. Dr David Gott stated that the UK has a long history of food safety assessment. The COT was established in 1978 to provide advice and views on the science. This replaced the toxicology sub-committee of the Committee on Medical Aspects of Chemicals in Food and the Environment. In 1978, the committee was largely concerned with the approval of food additives and ingredients such as enzymes and colours. Initially, the main activity was reviewing the safety in use of food colourings at the request of the Food Additives and Contaminants Committee (FACC). The COT report was annexed to the FACC report which was published in 1979.
151. Many of the first topics in the 1970s and 1980s are familiar and still ‘burning issues’ to us today such as assessments on sweeteners, implications of survey data and derivation/discussion of Acceptable Daily Intakes (ADIs).
152. By the late 1990s most of the regulated product assessments for the UK were undertaken by the EU (EFSA), there were a few food additives/enzymes reviewed by COT, and also mainly non-food issues. However, EFSA toxicity testing guidelines are still based on the original guidelines of 1991 and need to be updated.
153. The UK have to “be clear in what we think, why we think it and what we don’t know” as funded research moves fast and soon becomes outdated.
154. From the early 1990’s onwards, openness on COT advice became customary. With open meetings and workshops, and all papers published, ‘very little should need to be hidden’. The 1st Scientific Advisory Committee annual report was 30 years ago (1991), which included member details and declarations of interest. There is a willingness to work with the other committees and sub-committees to try and be as transparent in our advice as possible. Increased moves toward openness and transparency included working papers and minutes which are now published on the COT website and meetings are held in open session. The exceptions to this being where data are confidential through being commercially sensitive or pre-publication.
155. By 1998, most of the food approvals were taking place at an EU level and the COT reviewed only a few food additives and applications on a voluntary enzyme scheme. Most of the COT’s time was taken up with surveys, contaminants and non-food issues.
156. Highlights of the more recent past include collaboration with other advisory committees, risk benefit assessment of oily fish, early years and the maternal diet, caffeine in pregnancy and energy drinks.
157. The COT are now thinking about how data are handled using the approaches developed in the Synthesising Epidemiological Evidence Subgroup (SEES) and Synthesising Epidemiological and Toxicological Evidence (SETE) reports, as well as the COT and FSA NAMs roadmap. There is independent scrutiny of e.g., opinions, guidance, new methodological approaches from international (and national) risk assessment authorities. In future, quick decisions will need to be made using more limited data therefore, new methods and technologies will need to be embraced, to assist in this. There will also be a need to use mechanistic and kinetic data to strengthen the rationale. We will need to update guidance to reflect these changes.
158. There will be a need to embrace NAMs and potentially take some risk (compared to previous historical methods), as well as an opportunity for using artificial intelligence (AI) for e.g., literature searches. The guidance for both risk assessment and risk management will need to be updated to reflect this changing landscape.
159. The COT and FSA have currently based their scientific requirements on international guidance to provide certainty and consistency. There was a clear commitment to the use of sound science and risk-based approaches. There was clear communication with stakeholders, senior management and the SACs and other government departments what is known and what is not known and attempted to outline uncertainty and the limits of our advice.
160. Testing will have to get smarter to enhance the 3Rs principles. Implications of moving from apical to non-apical end points will need to be understood and more thinking around uncertainty factors. The question of small shifts in parameter ranges needs to be addressed and rethink adversity and variation. The main aim is still to protect humans but society and risk managers will need to decide to what extent. This is currently another period of change with new challenges. Therefore, it is important that we have consistent accurate risk assessments. Finally, any new tiered approach will have to be explained in detail with the context.
Professor Tim Gant (UKHSA) presented from a personal perspective on the opportunities of EU exit, the challenges and the needs of government to achieve.
161. Toxicology has evolved over 30 years with omics technologies, AOPs and 21st century toxicology.
162. Progress is being made with the use of AOPs, Quantitative structure–activity relationship (QSARs) and the first skin sensitisation test guideline. Molecular methods have and will continue to lead to new NAMs. To translate this academic knowledge into application, the right people need to have the right knowledge. Such people would need to understand both molecular toxicological science and regulation as well as the needs of government and the public. There is little training that covers all these areas currently available.
163. ECHA and EFSA are both governed by EU law but operate independently. Since leaving the EU the UK have lost the capability of independent advice that is provided by the JRC for the EU and in particular laboratory capabilities, with the exception of a few UK HSA laboratories. As stated by the RSC, there is a lack of a central research facility that matches the Institute for Health and Consumer Protection and the EU reference laboratory. We have some exemplary primary research work in CEFAS and UK HSA but it is resource limited and not well recognised by sponsoring departments. Whilst there is joining up at the scientific levels, unfortunately the science is largely supported by short term funding and has capacity issues. There is also a lack of facilities, as an example, a repository for materials.
164. Professor Gant outlined the breadth of work the UK HSA Toxicology department undertake, towards the prevention of health effects from environmental chemicals. This work is done collaboratively with cross government departments and universities and includes development of regulatory test methods, health protection, teaching and outreach and applied translational research.
165. Divergence from EU REACH might be expected in the following areas: endocrine disruptors; persistent mobile and toxic chemicals; methods of testing and in particular the use of NAMs; differential registrations in the authorised and restricted registers; a risk rather than hazard-based assessment; and mixtures and mixture assessment factors (MAF). Divergence in these areas could be beneficial for the UK post EU exit but it was emphasised that divergence cannot take place without robust scientific understanding and knowledge to inform policy. The capacity to develop this within either government or academia is limited and will hinder the exploitation of the opportunities that exist.
166. Therefore, the important take home message is that where there is going to be divergence, robust science will be required and much of this requirement will fall to UKHSA and other government departments. There is a need to ensure that the best scientific advice is available from within the UK or from the global scientific networks. How is the UK going to provide the evidence? A research capacity needs to be developed in addition to capability and capacity in validation research. There is currently no capacity in the UK for this and it is crucial to the application of NAMs, for example.
167. There need to be resilience in skill sets and succession planning to maintain corporate memory. Finally, government needs to work with academia and industry. We have opportunities; so, how are we going to create structures, and resources to lead and develop? It has to be science based and that requires collaborative working across all relevant sectors facilitated and led from government.
Next Generation Toxicology Training in the UK
Dr Sarah Judge presented on toxicology training in the UK.
168. Dr Sarah Judge presented the research which she has co-ordinated, looking at toxicology skills training and recruitment in the UK, which falls under the Education and Skills Gap Project of the British Toxicology Society. So far, phase 1 of this project has been completed which aimed to identify knowledge and skill requirements, as well as to assess current education and training provision and identify gaps.
169. In terms of phase 1, several expert representatives of the UK regulatory toxicology community were approached, which covered regulatory consultancies, product companies, contract research organisations (CROs) and UK government (including the FSA). This was done to get a consensus on the knowledge and skills required for entry into this job market and looking at intermediate and expert job roles now and 5 years from now.
170. The results from the survey showed a total of 751 knowledge and skills items across the whole regulatory toxicology sector, however as there was some overlap, this was consolidated to just 189 items. The next stage was for the representatives to rate each of these items as unnecessary, unimportant, important, or necessary for entry, intermediate, and expert level job roles. A consensus was acknowledged if there was over 70% agreement for each item. For example, for an entry level role, it is expected that under ‘dose response assessment’, that candidates understand how toxicology reference endpoints are derived.
171. Overall the results showed that with increasing seniority of job role, more knowledge and skills items are required. Also, CROs were shown to require the least number of items, whereas regulatory consultancies require the most knowledge and skills items.
172. Currently, the toxicology education and training provision in the UK is being assessed to identify any gaps, to ensure that graduates are suitably prepared for the regulatory toxicology job market, and in respect of CPD (Continuing Professional Development) activities for current employees.
173. The upcoming phases 2 and 3 of this project will look at facilitating the provision of education and training required for UK regulatory toxicology roles. For entry level jobs, Dr Judge suggested that this could involve collaborating with course leaders at UK universities to provide the list of entry level knowledge and skills that were identified under project 1. For intermediate & expert level jobs, Dr Sarah Judge suggested that this could involve collaborating with course leaders of current CPD courses by helping them meet items, as well as to improve quality control.
174. Overall, the talk highlighted the need to ensure that university graduates have the right knowledge and skills, and that current staff in regulatory toxicology are accessing CPD courses, and what is being done to ensure that the knowledge and skills items are achieved.
Roundtable Discussions-When to? How? what next?
The following questions were used as prompts for the discussion:
• Does the UK have the capacity and capability in human and environmental safety, for example in CRO’s and consultancies to support a different regulatory approach for example in the use of NAMs
• Should the UK take a more strategic approach to the provision of university education and training courses and programmes in regulatory toxicology to tackle the current skills shortage in chemical and food safety both in industry and government departments?
• What would a science-based, health protective approach to food and chemicals regulation in the UK look like in practice?
• Would the development and validation of new approaches to chemical and food safety assessment, including NAMs, benefit from the creation of a UK centre of excellence dedicated to this purpose?
Roundtable Discussions
Themes
175. Centre of Excellence: The question was raised ‘do we need a centre of excellence on risk assessment and science to be involved in research and training and if so, should we involve the chief scientific advisors (CSA) from the relevant bodies?’ It was questioned if the CSAs will actually join in to get the message across to senior managers and how do we get them to participate. It was highlighted that a significant change on how we do business is required and that CSAs should be aware as it would be a shame if we cannot engage CSAs as to how we can implement change. It was suggested that at regular meetings of the CSA we could get them to agree to participate, however a coherent case needs to be put together.
176. Next generation toxicology training: Another issue that was raised was that young people need to go into degrees wanting to become a toxicologist. Toxicology is not well rated in academia. It was noted that an increase in university students undertaking degrees with the intention of becoming toxicologists was required. It was suggested that if a centre of excellence was set up, a cluster of businesses could be set up around the centre and this could overcome the rating of toxicology. Another point raised was regarding the skills required for toxicologists at different levels of expertise. It was mentioned that it was difficult for any younger toxicologists to get on to the SACs.
177. A suggestion was raised as to whether the training of individuals, who are newer to the field, was classed as mentoring within the civil service. This could be achieved through protected time for more senior colleagues to pass on their knowledge to those below them to facilitate the intermediate skill development.
178. Funding: It was stated that one of the biggest issues was with training and lack of funding and it was suggested that the UK should become involved in tackling this. It was also highlighted that it may cost millions to achieve this goal but that there is value in investment to get economy GDP increased and make the UK competitive. One way to achieve this is to make the UK a centre of regulatory excellence.
It was suggested that the UK needs to become involved and funders are needed. It was questioned how we get senior management to participate in this. It was stated that if those in decision making positions can’t be engaged with, implementing change will be impossible. A suggested solution was to get the issue raised by CSAs at senior meetings. It was highlighted that there is currently no Science Minister but there is likely to be a new minster in September.
Concluding Thoughts and Forward Look
Figure 1. Workshop outputs and forward look.
179. Increase transparency with the public to ensure trust will be key: There needs to be a good balance of opportunities that may arise from a UK approach to make sure that there is no perception that there will be lower standards in regulation. The public needs to be engaged with to ensure trust that change in regulation is not a change in consumer protection. It needs to be explained clearly to the public what has been decided and why it has been decided.
180. Good quality decisions need to be taken: The question needs to be asked, what level of protection is required? And ensure that the evidence is used wisely. The UK needs to develop a systematic approach to risk assessment. The UK can build its own regulatory frameworks according to the best available evidence, ensuring that science meets regulatory needs.
181. The UK need to embrace new approaches: In future we will need to make quick decisions with limited or very limited data. This is an opportunity to use the best science available, for example through the use of new methodologies in hazard and risk assessment (NAMs). This will lead to more robust, reproducible, translatable, cost-effective, rapid and ethically acceptable assessment of chemicals.
182. The UK need to collaborate and harmonise: The government needs to work with academia and industry and vice versa. International alignment and collaboration is key.
183. The UK should expand its horizons: The UK need to learn from other international regulators and not be limited by a “not invented here” approach. The UK needs to continue to build its reputation as a world leader in the field.
184. The UK should train the next generation: A strategy needs to be developed and implemented to train the next generation of scientists and risk assessors, including but not limited to utilising alternative approaches and knowledge for risk assessment.
References
COT FSA UK Paving the way for a UK Roadmap: Development, Validation and Regulatory Acceptance of New Approach Methodologies (NAMs) in Chemical Risk Assessment Draft Version 2.
COT FSA UK NAMs Roadmap Draft Version 2 (2021) (food.gov.uk)
Doe, J.E., Boobis, A.R., Cohen, S.M. et al. The codification of hazard and its impact on the hazard versus risk controversy. Arch Toxicol 95, 3611–3621 (2021).
https://doi.org/10.1007/s00204-021-03145-6.
DEFRA 25-year environmental plan (2018)
25 Year Environment Plan - GOV.UK (www.gov.uk).
FSA Strategy: Food you can trust (2022)
Food you can trust.
HSE Strategy: Protecting people and places (2022)
Protecting people and places: HSE strategy 2022 to 2032.
Royal Society of Chemistry Policy Document : Drivers and scope for a UK chemicals framework (2019)
rsc-uk-chemical-framework-drivers-scope-2020.pdf.
Royal Society of Chemistry risk based policy position on EDCs (2020)
edc-policy-position-final-sep-2020.pdf (rsc.org)
Royal Society of Chemistry Risk-based regulation for per- and poly-fluoroalkyl substances (PFAS)
pfas-policy-position-dec-2021.pdf (rsc.org)
Regulating after EU exit Report- National Audit Office (2022)
Regulating after EU Exit - National Audit Office (NAO) report.
UK legislation
Legislation.gov.uk.
UK Government Genetic Bill
UK GOV Genetic Bill.
Abbreviations
|
Acceptable Daily Intakes |
ADIs |
|
Additives, Enzymes, and other Regulated products |
AER |
|
Advisory Committee on Animal Feeding stuffs |
ACAF |
|
Animal Food and Feed Additives |
AFFA |
|
Adverse outcome pathways |
AOPs |
|
Control of Substances Hazardous to Health |
COSHH |
|
Committee on the Toxicity of Chemicals in Food, Consumer Products and the Environment |
COT |
|
the Committee on Carcinogenicity of Chemicals in Food, Consumer Products and the Environment |
COC |
|
Committee on Mutagenicity of Chemicals in Food, Consumer Products and the Environment |
COM |
|
Contract research organisations |
CROs |
|
Department for Environment Food and Rural Affairs |
Defra |
|
Department of Health and Social Care |
DHSC |
|
European Food Safety Authority |
EFSA |
|
European Union |
EU |
|
US Environmental Protection Agency |
EPA |
|
Food Contact Materials |
FCM |
|
Food Standards Agency |
FSA |
|
Genetically modified organism |
GMO |
|
Health-based guidance values |
HBGVs |
|
Health and Safety Executive |
HSE |
|
Integrated Approaches to Testing and Assessment |
IATA |
|
Joint Expert Group |
JEG |
|
Mutual acceptance of data |
MAD |
|
Metabolic disrupting chemicals |
MDCs |
|
Mode of Action |
MOA |
|
Non intentionally added substances |
NIAS |
|
Next Generation Risk Assessment |
NGRA |
|
Organisation for Economic Co-operation and Development |
OECD |
|
Partnership for the assessment of risk from chemicals |
PARC |
|
Perfluoroalkyl and Polyfluoroalkyl Substances |
PFAS |
|
Quantitative structure–activity relationship |
QSARs |
|
Registration, Evaluation, Authorisation and Restriction of Chemicals |
REACH |
|
Royal Society of Chemistry |
RSC |
|
Scientific Advisory Committees |
SACs |
|
United Kingdom |
UK |
|
UK Health Security Agency |
UK HSA |
|
UN Environment Program |
UNEP |
Organizing Committee
Dr Olivia Osborne
Ms Cath Mulholland
Professor Tim Gant
Dr Phil Botham
Ms Sophy Wells
COT Members
Professor Alan Boobis (COT Chair)
Dr Phil Botham
Ms Jane Case
Dr Stella Cochrane
Dr James Coulson
Dr Silvia Gratz
Professor Thorhallur Ingi Halldórsson
Professor Gary Hutchison
Dr Sarah Judge
Professor Gunter Kuhnle
Dr David Lovell
Professor Shirley Price
Dr Mac Provan
Ms Juliet Rix
Dr Michael Routledge
Dr Cheryl Scudamore
Dr Natalie Thatcher
Professor Mireille Toledano
Dr Simon Wilkinson
Professor Philippe Wilson
Professor Matthew Wright
Professor Maged Younes
COT Secretariat
Ms Cath Mulholland -FSA Scientific Secretary
Ms Britta Gadeberg- UK HSA Scientific Secretary
Ms Natasha Adams
Ms Rhoda Aminu
Dr Alex Cooper
Dr Barbara Doerr
Mr Michael Dickinson
Dr Gail Drummond
Mr Lawrence Finn
Ms Emma French
Ms Jocelyn Frimpong-Manso
Dr David Gott
Mr Thomas Hornsby
Ms Cleanncy Hoppie
Dr Emily Hudson
Dr David Kovacic
Mr Barry Maycock
Dr Olivia Osborne
Ms Claire Potter
Dr Joseph Shavila
Dr Gaetana Spedalieri
Ms Sabrina Thomas
Ms Chara Tsoulli
Ms Frederique Uy
Ms Sophy Wells